Topics
>RESEARCH
Antifibrotic Drug Shows Promise for Premature Ovarian Insufficiency
Study suggests ovarian fibrosis may act as a mechanical barrier to follicle growth and can be reduced using an approved antifibrotic drug
Premature ovarian insufficiency (POI) affects 1–3% of women and has limited treatment options. Researchers report that ovarian fibrosis may restrict follicle development and that the antifibrotic drug finerenone reduced collagen deposition and promoted follicle growth in mice and in a small exploratory study of women with POI. While larger trials are needed, the findings suggest that repurposing antifibrotic therapy could offer a new, less invasive treatment approach.
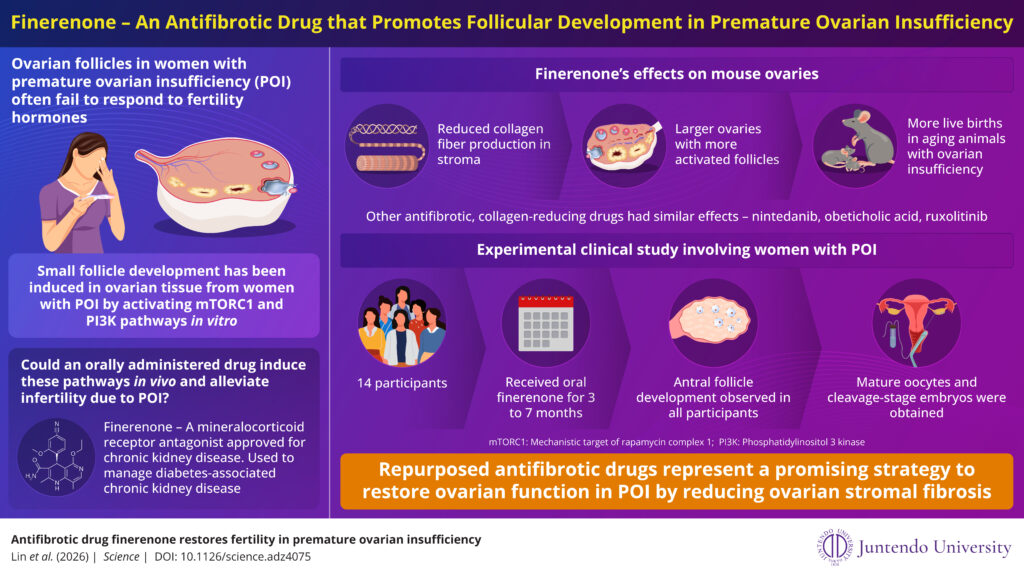
Image title: Finerenone – An antifibrotic drug that counters premature ovarian insufficiency
Image caption: Fibrosis in the ovarian cortex is a major barrier to follicle formation and is a mechanical cause of premature ovarian insufficiency. Finerenone reduces ovarian fibrosis and restores fertility
Image credit: Prof. Kazuhiro Kawamura from Juntendo University
License type: Original content
Usage restrictions: You are free to share and adapt the Infographic material, but attribution is required, with a link to the news source.
Premature ovarian insufficiency (POI) is a clinically significant cause of infertility that affects between 1 to 3% of women of childbearing age. Symptoms include absent menses, low estrogen levels, and elevated follicle-stimulating hormone (FSH) levels. Although residual primordial follicles often remain in the ovaries of women with POI, these follicles frequently fail to develop spontaneously. Because FSH levels are already high, the follicles typically do not respond to additional hormonal stimulation used in standard fertility treatments.
Researchers from Juntendo University, led by Professor Kazuhiro Kawamura, previously developed an invasive in-vitro technique to restore fertility in women with POI. Professor Kui Liu’s team from the University of Hong Kong (HKU) discovered that the mechanistic target of rapamycin complex 1 (mTORC1) pathway in granulosa cells and the phosphatidylinositol 3 kinase (PI3K) pathway in oocytes were critical to follicle formation. “Based on this knowledge, the in-vitro activation technique was developed to activate small follicles in cultured ovarian cortical tissues from patients with POI and then autotransplanted back into the patients under a laparoscopic surgery, resulting in documented pregnancies and live births,” said Prof. Kawamura.
Building on these mechanistic insights, the researchers investigated whether an already approved oral medication might stimulate follicle development without requiring invasive procedures. Prof. Kawamura notes that drugs that activate the mTORC1 and PI3K pathways are already used to treat kidney disorders. Could one such drug be repurposed to restore fertility in women with POI? Prof. Kawamura worked with Prof. Liu’s team to explore this concept. Zexiong Lin and Dr. Yuan Li of Prof. Liu’s team, and Dr. Tianren Wang of HKU – Shenzhen Hospital also contributed greatly to this research effort. Their findings were published on February 5, 2026, in Volume 391, Issue 6785 of the journal Science.
The team identified finerenone as a promising candidate. Finerenone is a mineralocorticoid receptor antagonist known for its antifibrotic properties and favorable safety profile in patients with chronic kidney disease. When ovaries from immature mice were grown in-vitro and dosed with finerenone, they began to develop follicles and eventually mature oocytes.
Encouraged by these findings, the team tested the effects of oral doses of finerenone on mouse fertility. Over an 18-week period, adult mice receiving finerenone gave birth to more offspring than those on a placebo. What’s more, finerenone also induced follicle formation in mice that had grown infertile due to ovarian insufficiency caused by aging.
Gene expression analysis showed that finerenone suppressed the production of collagen in the ovarian cortex. Excess collagen deposition contributes to tissue stiffening (fibrosis), which may physically restrict small follicle growth. “Finerenone’s antifibrotic effect alleviates ECM-mediated constraints on small follicle growth, thereby allowing follicles to develop. Furthermore, we identified the stromal collagen–granulosa signaling as an important negative regulator of follicular development,” said Prof. Kawamura, adding, “Therefore, finerenone stimulates follicular development through its antifibrotic action on the ovarian stroma.” Other drugs such as nintedanib and ruxolitinib also induced follicle formation in mouse ovaries. These drugs prevent fibrosis through a different mechanism from finerenone, which confirmed the collagen-induced constraint hypothesis.
Would these findings hold true in humans? The team enrolled 14 women with POI who were being treated at HKU Shenzhen Hospital for an experimental study. Participants received oral finerenone for a period of 3 to 7 months. Follicle development was seen in all participants. Seven patients produced mature oocytes that could be used for in-vitro fertilization (IVF). The quality of oocytes harvested was comparable to that of same-age women without POI undergoing IVF. While further trials are needed, these findings provide early evidence that antifibrotic therapy may enhance follicle activation in some women with POI.
Discussing these encouraging results, Prof. Kawamura concludes, “Further clinical characterization of FDA-approved oral antifibrotic drugs for their abilities in activating small ovarian follicles in patients offers a promising path to repurposing therapies for POI-related infertility.”
If validated in larger trials, antifibrotic therapy could represent a less invasive alternative to current experimental approaches and expand treatment options for women with POI worldwide.
***
Reference
|
Authors |
Zexiong Lin1,2, Yuan Li2, Yu Zhao2, Dongteng Liu1, Shuzi Deng1,2, Jingkai Gu2, Yanyan Li2, Xudong Zhao1, Peishan Wu2, Yuan Xiao1, Jiaping Su2, Yiting Sun1,2, Yihui Zhang1, Yin Lau Lee1, Yorino Sato3, Haitao Zeng4, Haonan Lu1, Juanhui Zhang2, Jennifer K.Y. Ko1, Jing Zhao2, Kazuhiro Kawamura3, Ernest H.Y. Ng1, Shanfang Jiang5, Yu Li2, Xi Xia6, Karen K.L. Chan1, William S.B. Yeung1,2, Tianren R. Wang1,2*, Kui Liu1,2* |
|
Title of original paper |
Antifibrotic drug finerenone restores fertility in premature ovarian insufficiency |
|
Journal |
Science, Volume 391, Issue 6785, 2026 |
|
DOI |
|
|
Affiliations |
1Department of Obstetrics and Gynecology, School of Clinical Medicine, Li Ka Shing Faculty of Medicine, The University of Hong Kong, Pokfulam, Hong Kong, China. 2Shenzhen Key Laboratory of Fertility Regulation, Center of Assisted Reproduction and Embryology, The University of Hong Kong-Shenzhen Hospital, Shenzhen, Guangdong, China. 3Department of Obstetrics and Gynecology, Juntendo University Faculty of Medicine, Tokyo, Japan. 4Center of Reproductive Medicine, The Sixth Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, China. 5Shenzhen Guangming District People’s Hospital, Shenzhen, China. 6Department of Reproductive Center, Peking University-Shenzhen Hospital, Shenzhen, Guangdong, China. |
***
About Professor Kazuhiro Kawamura from Juntendo University
Dr. Kazuhiro Kawamura is a Professor at the Department of Obstetrics and Gynecology, Faculty of Medicine, Juntendo University. Prof. Kawamura completed his graduate studies at Akita University and conducted postdoctoral research at Akita and Stanford Universities. His research focuses on ovarian physiology, reproductive endocrinology, and infertility. Prof. Kawamura has authored over 160 papers and 70 book chapters in these fields. He is the current director of the Japan Society for Reproductive Endocrinology and the Japan Society for Ova Research. He also serves on the boards of many prestigious national and international medical associations. He is one of the pioneers of infertility treatment of POI and has developed the in-vitro activation technique (IVA: in-vitro activation), which is now widely used for the treatment of POI.