Jun. 05, Mon, 2023
Topics
>RESEARCH
Patchwork of Mutations Contributing to Bipolar Disorder
Researchers find a link between bipolar disease and potentially pathogenic mosaic mutations found in neurodevelopmental disorder genes and mitochondrial tRNA genes
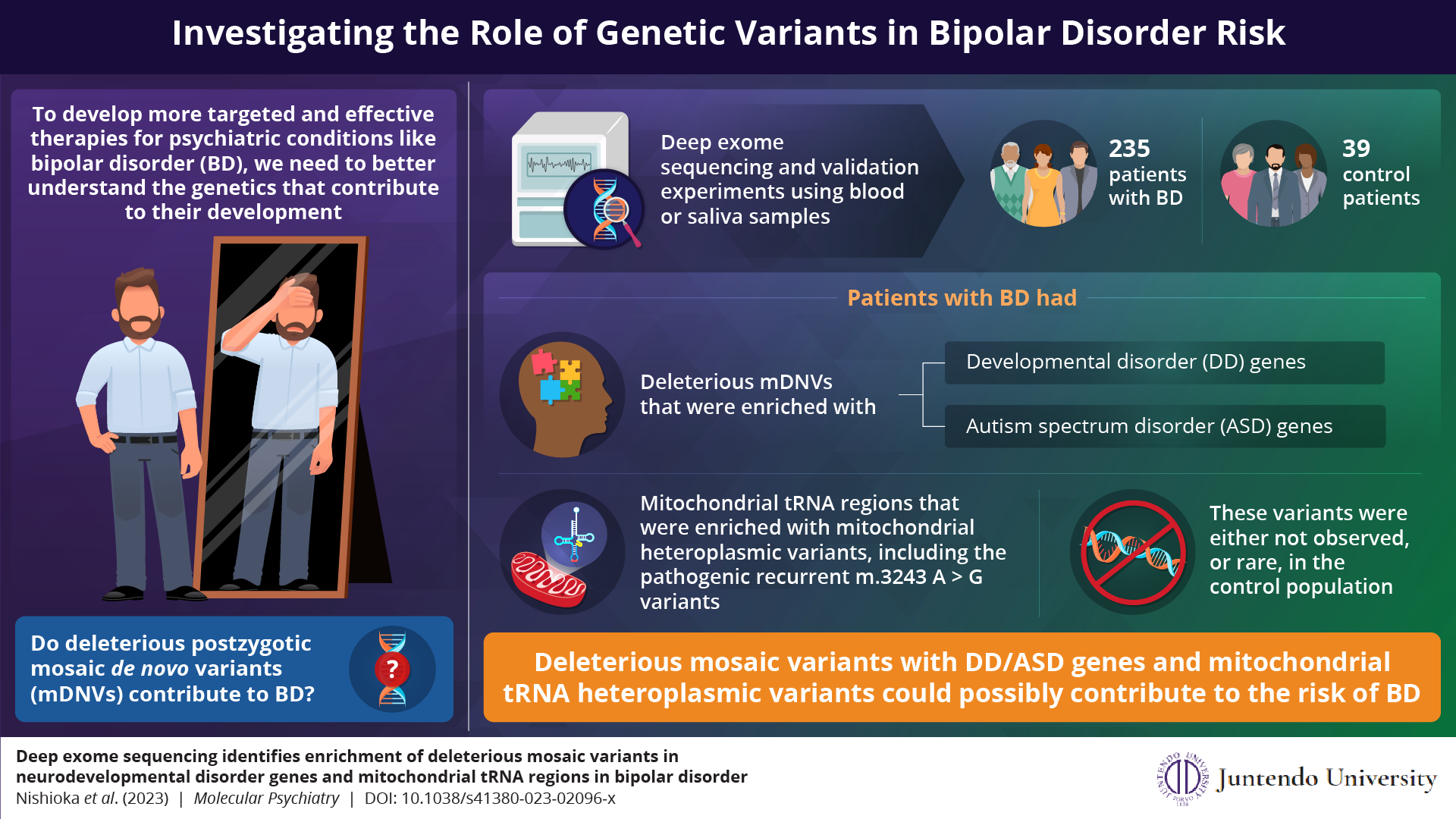 Japaneseresearchers showed that genes associated with developmental disorders and autism spectrum disorders are significantly enriched with deleterious mosaic mutations in patients with bipolar disorder (BD). Similarly, the mitochondrial tRNA region
of these patients also showed significant enrichment of deleterious mosaic mutations. These findings help better understand the genetics and pathogenesis of BD.
Japaneseresearchers showed that genes associated with developmental disorders and autism spectrum disorders are significantly enriched with deleterious mosaic mutations in patients with bipolar disorder (BD). Similarly, the mitochondrial tRNA region
of these patients also showed significant enrichment of deleterious mosaic mutations. These findings help better understand the genetics and pathogenesis of BD.
Image credits:Masaki Nishioka from Juntendo University School of Medicine
License type:Original content
Usage restrictions: You are free to share and adapt the Infographic material but attribution is required, with a link to the news source.
Bipolar disorder (BD) is a major psychiatric condition that afflicts about 1% of people. Symptoms of BD include sudden onset of depressive mood with loss of interest which alternates with a manic state of hyperactivity. The suffering of the patients and
societal cost of this disorder requires the use of continued therapeutic management. Current medications—although vital for patients with BD—are not perfect solutions, given their potential side-effects and treatment resistance. This necessitates the
development of better therapeutics for BD, including precision medicine. A major hindrance to this process, however, lies in our limited understanding of the underlying biological mechanisms of BD, i.e., its pathogenesis and the genetic architecture of
people with BD. Several studies have linked BD with hereditary mutations, but recent genomic studies are now focusing on somatic mosaic variants—new mutations occurring during developmental stages—as another possible mechanism behind psychiatric disorders
like BD.
In a new study published in Molecular Psychiatry on May 30, 2023, a team of researchers led by Associate Professor Masaki Nishioka of Juntendo University, Japan, investigated the association between mosaic variants and the risk of BD. The research team included Dr. Tadafumi Kato, also from Juntendo University, and Dr. Atsushi Takata from RIKEN Center for Brain Science. “Most analyses exploring the genetic mechanisms of BD involve extracting information from mutations that are shared among all the cells of the patients. However, mosaic de novo mutations or somatic mutations, which arise during development, are not shared among all the cells. We know very little about how these mutations influence diseases like BD. Therefore, for our study, we hypothesized that deleterious mosaic de novo variants (mDNVs) in the genes associated with developmental disorders may have a role in BD’s pathology,” explains Dr. Nishioka.
The team recruited 235 participants with BD and 39 control participants without psychiatric disorders. They collected blood or saliva samples from the participants and analyzed the DNA extracted from these samples using deep exome sequencing (DES) to detect mosaic variants that originated during early development. Participants with BD had mosaic variants enriched in genes that are responsible for causing developmental disorders (DD) and autism spectrum disorder (ASD). Moreover, the proteins encoded by the DD/ASD genes with the proteins of the mosaic variants were closely linked and had more protein-protein interactions than expected.
Surprisingly, the team also found significant heteroplasmic mutations (another class of mosaic variants) in mitochondrial tRNA genes of participants with BD. For reference, some tRNA mutations are known to be pathogenic for other diseases. In fact, two participants with mitochondrial tRNA mutations had recurrent m.3243 A > G variants, which are known to be major causal variants for mitochondrial diseases, MELAS, which is a serious neurodevelopmental disorder. This finding complements other studies that have found that patients with mitochondrial diseases often exhibit symptoms of bipolar disorder or schizophrenia.
Furthermore, both the sets of deleterious mosaic variants—mDNVs and mitochondrial tRNA variants—were either absent or rarely observed in the control participants. These results indicate that the molecular mechanisms underlying DD/ASD could also contribute to BD in a compromised way through mosaic mutations. Moreover, they suggest that mitochondrial tRNA variants could be associated with BD despite the patient showing no obvious symptoms of mitochondrial diseases.
With this study, the researchers demonstrate that mosaic mutations, particularly those in neurodevelopmental disorder genes and mitochondrial tRNA genes, may be involved in the pathophysiology of BD. Dr. Nishioka is encouraged by what their study’s findings mean for scientists pursuing the research of molecular pathologies in neuropsychiatric diseases. He concludes, “Our research sheds new light on the genetic architecture of BD and provides more insights into the pathological contribution of mosaic variants in human diseases. This could potentially pave the way and expedite new research for the development of more effective, precision medications for treating BD and other psychiatric disorders.”
About Associate Professor Masaki Nishioka from Juntendo University
Dr. Masaki Nishioka earned his M.D. and Ph.D. from The University of Tokyo. Since 2018, he has been a Research Scientist at the RIKEN Center for Brain Science and is an Associate Professor in the Department of Psychiatry at Juntendo University, Japan. He has published 15 peer-reviewed articles since 2012 and explores how somatic mutation analysis and the nervous structure of the brain can help explain mental disorders, behavior, and cognition.
Bipolar disorder (BD) is a serious psychiatric disorder affecting around 1% of the population worldwide. While medications are indispensable for these patients, they are not completely effective, and the underlying genetic architecture and pathogenesis of BD remain unresolved. Researchers from Japan have now used deep exome sequencing to show that mosaic variants/mutations found in genes related to developmental disorders and autism spectrum disease and in the mitochondrial tRNA region could be associated with BD.
Mosaic mutations in neurodevelopmental disorder genes and mitochondrial tRNA genes have been found to be involved in the pathophysiology of bipolar disorder
Mosaic mutations in neurodevelopmental disorder genes and mitochondrial tRNA genes have been found to be involved in the pathophysiology of bipolar disorder
 Japaneseresearchers showed that genes associated with developmental disorders and autism spectrum disorders are significantly enriched with deleterious mosaic mutations in patients with bipolar disorder (BD). Similarly, the mitochondrial tRNA region
of these patients also showed significant enrichment of deleterious mosaic mutations. These findings help better understand the genetics and pathogenesis of BD.
Japaneseresearchers showed that genes associated with developmental disorders and autism spectrum disorders are significantly enriched with deleterious mosaic mutations in patients with bipolar disorder (BD). Similarly, the mitochondrial tRNA region
of these patients also showed significant enrichment of deleterious mosaic mutations. These findings help better understand the genetics and pathogenesis of BD.Image credits:Masaki Nishioka from Juntendo University School of Medicine
License type:Original content
Usage restrictions: You are free to share and adapt the Infographic material but attribution is required, with a link to the news source.
In a new study published in Molecular Psychiatry on May 30, 2023, a team of researchers led by Associate Professor Masaki Nishioka of Juntendo University, Japan, investigated the association between mosaic variants and the risk of BD. The research team included Dr. Tadafumi Kato, also from Juntendo University, and Dr. Atsushi Takata from RIKEN Center for Brain Science. “Most analyses exploring the genetic mechanisms of BD involve extracting information from mutations that are shared among all the cells of the patients. However, mosaic de novo mutations or somatic mutations, which arise during development, are not shared among all the cells. We know very little about how these mutations influence diseases like BD. Therefore, for our study, we hypothesized that deleterious mosaic de novo variants (mDNVs) in the genes associated with developmental disorders may have a role in BD’s pathology,” explains Dr. Nishioka.
The team recruited 235 participants with BD and 39 control participants without psychiatric disorders. They collected blood or saliva samples from the participants and analyzed the DNA extracted from these samples using deep exome sequencing (DES) to detect mosaic variants that originated during early development. Participants with BD had mosaic variants enriched in genes that are responsible for causing developmental disorders (DD) and autism spectrum disorder (ASD). Moreover, the proteins encoded by the DD/ASD genes with the proteins of the mosaic variants were closely linked and had more protein-protein interactions than expected.
Surprisingly, the team also found significant heteroplasmic mutations (another class of mosaic variants) in mitochondrial tRNA genes of participants with BD. For reference, some tRNA mutations are known to be pathogenic for other diseases. In fact, two participants with mitochondrial tRNA mutations had recurrent m.3243 A > G variants, which are known to be major causal variants for mitochondrial diseases, MELAS, which is a serious neurodevelopmental disorder. This finding complements other studies that have found that patients with mitochondrial diseases often exhibit symptoms of bipolar disorder or schizophrenia.
Furthermore, both the sets of deleterious mosaic variants—mDNVs and mitochondrial tRNA variants—were either absent or rarely observed in the control participants. These results indicate that the molecular mechanisms underlying DD/ASD could also contribute to BD in a compromised way through mosaic mutations. Moreover, they suggest that mitochondrial tRNA variants could be associated with BD despite the patient showing no obvious symptoms of mitochondrial diseases.
With this study, the researchers demonstrate that mosaic mutations, particularly those in neurodevelopmental disorder genes and mitochondrial tRNA genes, may be involved in the pathophysiology of BD. Dr. Nishioka is encouraged by what their study’s findings mean for scientists pursuing the research of molecular pathologies in neuropsychiatric diseases. He concludes, “Our research sheds new light on the genetic architecture of BD and provides more insights into the pathological contribution of mosaic variants in human diseases. This could potentially pave the way and expedite new research for the development of more effective, precision medications for treating BD and other psychiatric disorders.”
About Associate Professor Masaki Nishioka from Juntendo University
Dr. Masaki Nishioka earned his M.D. and Ph.D. from The University of Tokyo. Since 2018, he has been a Research Scientist at the RIKEN Center for Brain Science and is an Associate Professor in the Department of Psychiatry at Juntendo University, Japan. He has published 15 peer-reviewed articles since 2012 and explores how somatic mutation analysis and the nervous structure of the brain can help explain mental disorders, behavior, and cognition.
Reference
| Authors | Masaki Nishioka 1,2,3,4, Jun Takayama5,6,7, Naomi Sakai1,3, An-a Kazuno3,4, Mizuho Ishiwata1,3, Junko Ueda3,4, Takashi Hayama8, Kumiko Fujii9, Toshiyuki Someya10, Shinichi Kuriyama11,12, Gen Tamiya5,6,7, Atsushi Takata 3,4,13 and Tadafumi Kato1,2,3 |
| Title of original paper | Deep exome sequencing identifies enrichment of deleterious mosaic variants in neurodevelopmental disorder genes and mitochondrial tRNA regions in bipolar disorder |
| Journal | Molecular Psychiatry |
| DOI | 10.1038/s41380-023-02096-x |
| Affiliations | 1 Department of Psychiatry and Behavioral Science, Juntendo University Graduate School of Medicine, Japan.
2 Department of Molecular Pathology of Mood Disorders, Juntendo University Graduate School of Medicine, Japan. 3 Laboratory for Molecular Dynamics of Mental Disorders, RIKEN Center for Brain Science, Japan. 4 Laboratory for Molecular Pathology of Psychiatric Disorders, RIKEN Center for Brain Science, Japan. 5 Department of AI and Innovative Medicine, Tohoku University School of Medicine, Japan. 6 Department of Integrative Genomics, Tohoku Medical Megabank Organization, Japan. 7 Statistical Genetics Team, RIKEN Center for Advanced Intelligence Project, Japan. 8 Yokohama Mental Clinic Totsuka, Japan. 9 Department of Psychiatry, Shiga University of Medical Science, Japan. 10 Department of Psychiatry, Niigata University Graduate School of Medical and Dental Sciences, Japan. 11 Department of Preventive Medicine and Epidemiology, Tohoku Medical Megabank Organization, Tohoku University, Japan. 12 Department of Molecular Epidemiology, Tohoku University School of Medicine, Japan. 13 Research Institute for Diseases of Old Age, Juntendo University Graduate School of Medicine, Japan. |