Mar. 03, Fri, 2023
Topics
>RESEARCH
Researchers Create Mutant Mice to Study Bipolar Disorder
Mice lacking FADS1/2—genes which have been associated with bipolar disorder—show behavioral swings between low and high activity phases.
 Researchers have created mutant mice that exhibit bipolar swings in activity, mirroring depressive and manic episodes of bipolar disorder in humans.
Image credits: Tadafumi Kato from Juntendo University
Researchers have created mutant mice that exhibit bipolar swings in activity, mirroring depressive and manic episodes of bipolar disorder in humans.
Image credits: Tadafumi Kato from Juntendo University
Image license: Original content
Bipolar disorder (BD) is a debilitating condition characterized by alternating states of depression (known as depressive episodes) and abnormal excitement or irritability (known as manic episodes). Large-scale genome-wide association studies (GWASs) have
revealed that variations in the genes present on the fatty acid desaturase (FADS) locus are linked to an increased risk of BD. Enzymes coded by FADS genes—FADS1 and FADS2—convert or “biosynthesize” omega-3 fatty acids into the different
forms required by the human body. Omega-3 fatty acids like eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) are crucial for the brain to function, and a reduction in the synthesizing activity of these molecules seems to increase susceptibility
to bipolar mood swings.
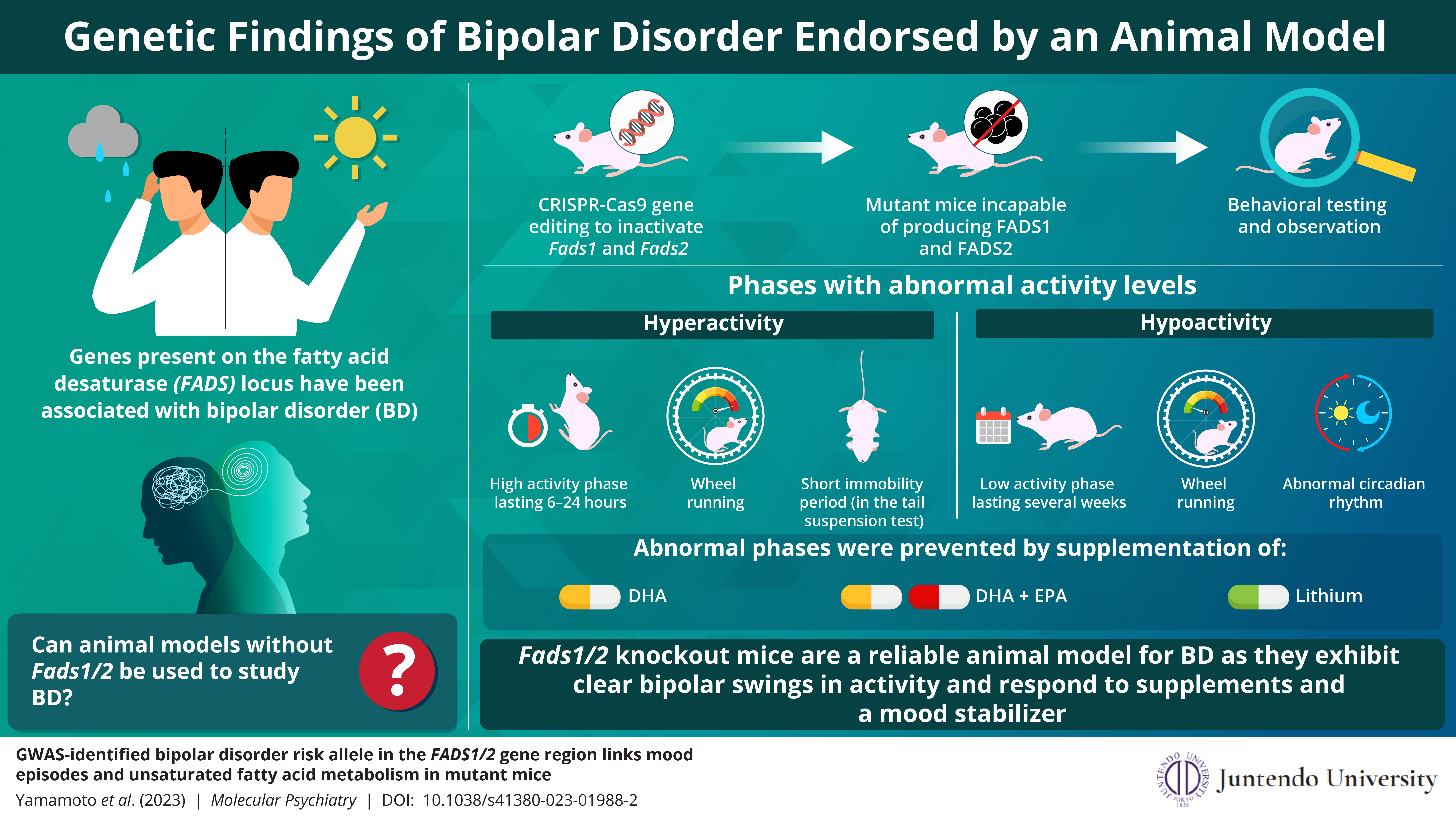
Research on most diseases involves establishment of an animal model of the disease. So, keeping this knowledge in mind, a team of researchers including Dr. Takaoki Kasahara and Hirona Yamamoto from RIKEN Brain Science Institute and Dr. Tadafumi Kato from Juntendo University in Japan, used CRISPR-Cas9 gene editing to create mutant mice that lack both Fads1 and Fads2 genes. They then tested these models to see if they could serve as an animal model for BD. Their tests involved behavioral analyses and systematic observation of these mice over six months, to identify behavioral changes caused by the mutation. Their findings are detailed in a study published on 21 February 2023 in Molecular Psychiatry.
The mice showed bipolar swings in behavior, featuring phases of abnormally low activity and phases of hyperactivity. Dr. Kato tells us, “The hyperactivity episodes, in which activity was far above the norm, usually lasted half a day“. During these episodes, the mice spent an increased amount of time running on wheels and were considerably less likely to remain still during tail suspension tests. Immobility during tail suspension tests is a widely used measure for depression-like behavior in mice.
Phases of low activity or “hypoactivity”, in which the mice exhibited low physical activity and spent an abnormally short period of time running on wheels, lasted much longer in the mutants—usually several weeks. “Unlike other locomotor activities, wheel running in mice is a strongly goal-directed behavior having a significant reward value; thus, a reduction in wheel running is associated with markedly diminished pleasure—anhedonia—a core symptom of a depressive episode,” Dr. Kato explains. Mutant mice also exhibited abnormal circadian rhythms during hypoactivity episodes. Circadian rhythm is the cyclical pattern of physiological changes that normally coincides with daytime and night-time; it affects our sleep pattern.
To ascertain whether behavioral changes can be reversed by increasing the levels of omega-3 fatty acids, the researchers fed mutant mice supplements containing DHA or a combination of DHA and EPA. The occurrence of periods with abnormally low activity in mice reduced considerably when EPA and DHA were supplemented. Administration of lithium, a mood stabilizer that is widely used to treat manic and depressive episodes in bipolar patients, had a similar effect on mutant mice. All of these indicate how similar BD in these mice models was to BD seen in humans.
Further, the researchers observed mice that lacked FADS enzymes exclusively in the brain to check if their behavioral changes can be attributed to molecular changes in brain cells. However, these mice failed to show bipolar swings in activity similar to mice that lacked FADS completely.
The study’s findings suggest that mutant mice lacking Fads1/2 may be helpful as an animal model to study the molecular roots of BD and the development and regulation of mood swings seen in the disorder. Moreover, since these mice respond positively to omega-3 fatty acid supplements and a mood stabilizer, they may help develop new treatments for BD.
Large-scale genome-wide association studies (GWASs) have suggested the involvement of genes present at the fatty acid desaturase (FADS) locus, and two of the enzymes they encode—FADS1 and 2—in regulating bipolar disorder. Now, researchers have found that mutant mice lacking Fads1/2 exhibit bipolar swings in behavior, suggesting that they might be useful as animal models to study bipolar disorder.
A New Mouse Model to Study Bipolar Disorder
A New Mouse Model to Study Bipolar Disorder
 Researchers have created mutant mice that exhibit bipolar swings in activity, mirroring depressive and manic episodes of bipolar disorder in humans.
Image credits: Tadafumi Kato from Juntendo University
Researchers have created mutant mice that exhibit bipolar swings in activity, mirroring depressive and manic episodes of bipolar disorder in humans.
Image credits: Tadafumi Kato from Juntendo University
Image license: Original content
Research on most diseases involves establishment of an animal model of the disease. So, keeping this knowledge in mind, a team of researchers including Dr. Takaoki Kasahara and Hirona Yamamoto from RIKEN Brain Science Institute and Dr. Tadafumi Kato from Juntendo University in Japan, used CRISPR-Cas9 gene editing to create mutant mice that lack both Fads1 and Fads2 genes. They then tested these models to see if they could serve as an animal model for BD. Their tests involved behavioral analyses and systematic observation of these mice over six months, to identify behavioral changes caused by the mutation. Their findings are detailed in a study published on 21 February 2023 in Molecular Psychiatry.
The mice showed bipolar swings in behavior, featuring phases of abnormally low activity and phases of hyperactivity. Dr. Kato tells us, “The hyperactivity episodes, in which activity was far above the norm, usually lasted half a day“. During these episodes, the mice spent an increased amount of time running on wheels and were considerably less likely to remain still during tail suspension tests. Immobility during tail suspension tests is a widely used measure for depression-like behavior in mice.
Phases of low activity or “hypoactivity”, in which the mice exhibited low physical activity and spent an abnormally short period of time running on wheels, lasted much longer in the mutants—usually several weeks. “Unlike other locomotor activities, wheel running in mice is a strongly goal-directed behavior having a significant reward value; thus, a reduction in wheel running is associated with markedly diminished pleasure—anhedonia—a core symptom of a depressive episode,” Dr. Kato explains. Mutant mice also exhibited abnormal circadian rhythms during hypoactivity episodes. Circadian rhythm is the cyclical pattern of physiological changes that normally coincides with daytime and night-time; it affects our sleep pattern.
To ascertain whether behavioral changes can be reversed by increasing the levels of omega-3 fatty acids, the researchers fed mutant mice supplements containing DHA or a combination of DHA and EPA. The occurrence of periods with abnormally low activity in mice reduced considerably when EPA and DHA were supplemented. Administration of lithium, a mood stabilizer that is widely used to treat manic and depressive episodes in bipolar patients, had a similar effect on mutant mice. All of these indicate how similar BD in these mice models was to BD seen in humans.
Further, the researchers observed mice that lacked FADS enzymes exclusively in the brain to check if their behavioral changes can be attributed to molecular changes in brain cells. However, these mice failed to show bipolar swings in activity similar to mice that lacked FADS completely.
The study’s findings suggest that mutant mice lacking Fads1/2 may be helpful as an animal model to study the molecular roots of BD and the development and regulation of mood swings seen in the disorder. Moreover, since these mice respond positively to omega-3 fatty acid supplements and a mood stabilizer, they may help develop new treatments for BD.
Reference
| Authors | Hirona Yamamoto1,2,3, Hyeon-Cheol Lee-Okada4, Masashi Ikeda5, Takumi Nakamura2,6, Takeo Saito5, Atsushi Takata2,7, Takehiko Yokomizo4, Nakao Iwata5, Tadafumi Kato1,6* and Takaoki Kasahara1,8,9,10* |
| Title of original paper | GWAS-identified bipolar disorder risk allele in the FADS1/2 gene region links mood episodes and unsaturated fatty acid metabolism in mutant mice |
| Journal | Molecular Psychiatry |
| DOI | 10.1038/s41380-023-01988-2 |
| Affiliations | 1Laboratory for Molecular Dynamics of Mental Disorders, RIKEN Center for Brain Science, Japan. 2Laboratory for Molecular Pathology of Psychiatric Disorders, RIKEN Center for Brain Science, Japan. 3Department of Neuropsychiatry, Graduate School of Medicine, The University of Tokyo, Japan. 4Department of Biochemistry, Juntendo University School of Medicine, Japan. 5Department of Psychiatry, Fujita Health University School of Medicine, Japan. 6Department of Psychiatry and Behavioral Science, Juntendo University Graduate School of Medicine, Japan. 7Research Institute for Disease of Old Age, Juntendo University School of Medicine, Japan. 8Career Development Program, RIKEN Center for Brain Science, Japan. 9Neurodegenerative Disorders Collaboration Laboratory, RIKEN Center for Brain Science, Japan. 10Present address: Institute of Biology and Environmental Sciences, Carl von Ossietzky University of Oldenburg, Germany. |