May. 30, Tue, 2023
Topics
>RESEARCH
A Novel Blood Serum Assay to Diagnose Neurodegenerative Diseases
Scientists have developed a new assay that can identify specific neurodegenerative diseases by detecting abnormal α-synuclein in patient sera
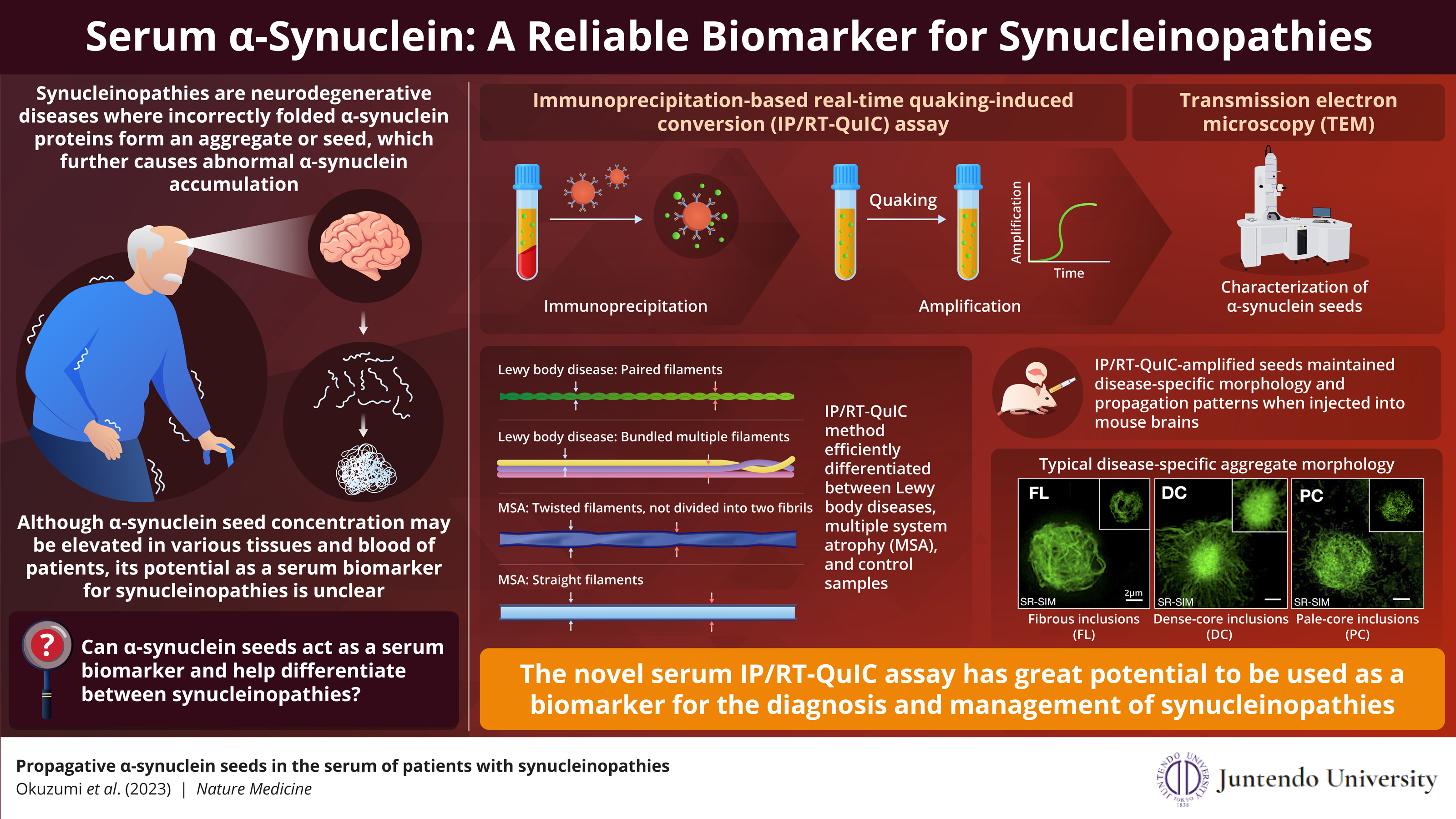 The IP/RT-QuIC assay efficiently detects minute concentrations of serum α-synuclein seeds, enabling quicker diagnosis of specific neurodegenerative disorders.
The IP/RT-QuIC assay efficiently detects minute concentrations of serum α-synuclein seeds, enabling quicker diagnosis of specific neurodegenerative disorders.
Image credits:Professor Hattori and his team from Juntendo University School of Medicine
License type:Original content
Usage restrictions:Cannot be reused without permission
Synucleinopathies are a group of neurodegenerative diseases caused by the abnormal accumulation α-synuclein, a protein normally found in the brain and neurons. Incorrect folding of α-synuclein leads to formation of ‘seeds’, which attract more α-synuclein
proteins to form larger clumps. Although, α-synuclein seeds have been found in various tissues and blood of patients with synucleinopathies, its potential as a biomarker is ambiguous.
Recently, in a study published in Nature Medicine, Associate Professor Ayami Okuzumi along with Senior Associate Professor Taku Hatano, both from the Juntendo University School of Medicine, Senior Assistant Professor Gen Matsumoto at the Nagasaki University School of Medicine, and Professor Nobutaka Hattori from Juntendo University Faculty of Medicine /RIKEN Center for Brain Science, present a novel assay that can efficiently detect α-synuclein seeds from a patient’s serum.
In this assay, named immunoprecipitation-based real-time quaking-induced conversion (IP/RT-QuIC), the α-synuclein seeds are isolated from the patient’s serum through immunoprecipitation (protein separation using an antibody binding only to the target protein) followed by rapid amplification by real-time quaking-induced conversion (amplification induced by vigorous shaking). This method is highly sensitive, as it can detect serum α-synuclein seed concentrations as small as 1000pg/ml. This comes as great news since most existing diagnostic methods require cerebrospinal fluid for synuclein detection. The current study was made available/published on May 30, 2023.
Sharing the objective of their study, Professor Hattori and his team explains, “In this study, we validated the usefulness of our novel assay system, IP/RT-QuIC, as a diagnostic marker of synucleinopathies. We propose that the fibril morphology of serum α-synuclein seeds and aggregates derived by IP/RT-QuIC can discriminate between Parkinson’s disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA).”
The research team demonstrated that IP/RT-QuIC detected α-synuclein seeds efficiently in patients with neurodegenerative diseases and could distinguish them from people without degenerative diseases (controls). Next, they studied the structural properties of the amplified seeds using transmission electron microscopy (TEM). They observed that the synuclein seed structure varied with the type of synucleinopathy. PD and DLB seeds showed paired filaments whereas MSA seeds had two distinct structures – twisted and straight filaments. This finding was confirmed that IP/RT-QuIC coupled with TEM can differentiate between synucleinopathies based on disease specific seed structure.
Further, when the researchers transduced amplified seeds into the HEK293T cell line stably expressing GFP-fused human α-synuclein with p.A53T mutation (in vitro) and injected seeds into mouse brains (in vivo), the seeds retained their aggregate forming capacity and diseases-specific seed structure. These aggregates displayed different morphologies depending on the disease type. Thus, specific synucleinopathies can be diagnosed by IP/RT-QuIC from the structural differences of the α-synuclein seeds and their aggregates.
This technique could help provide a quick and efficient diagnosis to patients. Professor Hattori and his team explains, “At present, a neurologist’s consultation is necessary to diagnose synucleinopathies. However, using IP/RTQuIC, a general internist can make the diagnosis. Therefore, more patients with synucleinopathies may be diagnosed with precision and could receive appropriate treatment at an earlier stage.”
The authors conclude with their future vision, “Our new IP/RT-QuIC assay may have many future applications as a biomarker for precise diagnosis and monitoring of treatment of neurodegenerative diseases in clinical trials. This simple diagnostic method will enable establishment of personalized therapy options for synucleinopathies.”
Funding
This study was supported by AMED under Grant Number JP19dm0207070 (Brain Mapping by Integrated Neurotechnologies for Disease Studies) to N.H., JP19dm0107156 (Strategic Research Program for Brain Sciences) to T.H. and A.O., JP21wm0425015 (Brain Mechanisms and Integrated Technologies for Mental Health and Disease Studies) to T.H. and A.O., JP19ak0101112 (Research on Development of New Drugs) to T.H. and N.H., JP21dk0207055 (Research and Development Grants for Dementia) to T.H., and JP16dm0107140 (Strategic Research Program for Brain Sciences) to N.N.; Grants-in-Aid for Scientific Research (21H04820 to NH, 21K07424 to T.H., 19K16928 to A.O., and 20K22693 to SU) from the Japan Society for the Promotion of Science; Visionary Council on the Moonshot Research and Development Program (JPMJMS2024-5 to N.H.) grants-in-aid from the Research Committee of CNS Degenerative Disease, Research on Policy Planning and Evaluation for Rare and Intractable Diseases, Health, Labor, and Welfare Sciences Research Grants; the Ministry of Health, Labor, and Welfare, Japan to N.H.; The Setsuro Fujii Memorial Osaka Foundation for Promotion of Fundamental Medical Research to T.H.; and Grants-in-Aid from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) of Japan to GM (17K07098 and 20H05333) for Scientific Research on Innovative Areas “Multimode autophagy.” The current work was also supported by the National Centre of Excellence in Research on Parkinson’s Disease (NCER-PD) funded by the Luxembourg National Research Fund (FNR; NCER13/BM/11264123) and the PEARL program (FNR; PEARL /P13/6682797 to RK and FNR and PEARL P16/BM/11192868 to MM).
Contact
For inquiries, please contact:
In relation to this study
Department of Neurology, Juntendo University School of Medicine Nobutaka Hattori, Professor, E-mail: nhattori “AT” juntendo.ac.jp
Taku Hatano, Senior Associate Professor, E-mail: thatano“AT”juntendo.ac.jp
TEL: +81-3-3813-3111
About the press release
Juntendo University School of Medicine
Yoshitaka Nakashima, E-mail: y.nakashima.gd “AT” juntendo.ac.jp
TEL: +81-3-5802-1590
Matters related to AMEDs projects
Division of Basic Medical Research, Department of Basic Medical Research
E-mail: brain-pm”AT”amed.go.jp
Telephone: +81-3-6870-2286
Replace “AT” with “@”
Clusters of abnormally folded α-synuclein proteins, called seeds, trigger the formation of larger aggregates, leading to neurodegenerative disorders known as synucleinopathies. Researchersfrom Japanhave developed a novel assay that can detect α-synuclein seeds in the blood. This new assay is highly sensitive, simple, quick, and can differentiate between synucleinopathies based on structural differences of the amplified seeds. This research was supported by the National Research Program for Neurological Disorders and Mental Health of the Japan Agency for Medical Research and Development (AMED).
α-Synuclein seeds in patients’ sera are a reliable biomarker for diagnosis of synucleinopathies
α-Synuclein seeds in patients’ sera are a reliable biomarker for diagnosis of synucleinopathies
 The IP/RT-QuIC assay efficiently detects minute concentrations of serum α-synuclein seeds, enabling quicker diagnosis of specific neurodegenerative disorders.
The IP/RT-QuIC assay efficiently detects minute concentrations of serum α-synuclein seeds, enabling quicker diagnosis of specific neurodegenerative disorders.Image credits:Professor Hattori and his team from Juntendo University School of Medicine
License type:Original content
Usage restrictions:Cannot be reused without permission
Recently, in a study published in Nature Medicine, Associate Professor Ayami Okuzumi along with Senior Associate Professor Taku Hatano, both from the Juntendo University School of Medicine, Senior Assistant Professor Gen Matsumoto at the Nagasaki University School of Medicine, and Professor Nobutaka Hattori from Juntendo University Faculty of Medicine /RIKEN Center for Brain Science, present a novel assay that can efficiently detect α-synuclein seeds from a patient’s serum.
In this assay, named immunoprecipitation-based real-time quaking-induced conversion (IP/RT-QuIC), the α-synuclein seeds are isolated from the patient’s serum through immunoprecipitation (protein separation using an antibody binding only to the target protein) followed by rapid amplification by real-time quaking-induced conversion (amplification induced by vigorous shaking). This method is highly sensitive, as it can detect serum α-synuclein seed concentrations as small as 1000pg/ml. This comes as great news since most existing diagnostic methods require cerebrospinal fluid for synuclein detection. The current study was made available/published on May 30, 2023.
Sharing the objective of their study, Professor Hattori and his team explains, “In this study, we validated the usefulness of our novel assay system, IP/RT-QuIC, as a diagnostic marker of synucleinopathies. We propose that the fibril morphology of serum α-synuclein seeds and aggregates derived by IP/RT-QuIC can discriminate between Parkinson’s disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA).”
The research team demonstrated that IP/RT-QuIC detected α-synuclein seeds efficiently in patients with neurodegenerative diseases and could distinguish them from people without degenerative diseases (controls). Next, they studied the structural properties of the amplified seeds using transmission electron microscopy (TEM). They observed that the synuclein seed structure varied with the type of synucleinopathy. PD and DLB seeds showed paired filaments whereas MSA seeds had two distinct structures – twisted and straight filaments. This finding was confirmed that IP/RT-QuIC coupled with TEM can differentiate between synucleinopathies based on disease specific seed structure.
Further, when the researchers transduced amplified seeds into the HEK293T cell line stably expressing GFP-fused human α-synuclein with p.A53T mutation (in vitro) and injected seeds into mouse brains (in vivo), the seeds retained their aggregate forming capacity and diseases-specific seed structure. These aggregates displayed different morphologies depending on the disease type. Thus, specific synucleinopathies can be diagnosed by IP/RT-QuIC from the structural differences of the α-synuclein seeds and their aggregates.
This technique could help provide a quick and efficient diagnosis to patients. Professor Hattori and his team explains, “At present, a neurologist’s consultation is necessary to diagnose synucleinopathies. However, using IP/RTQuIC, a general internist can make the diagnosis. Therefore, more patients with synucleinopathies may be diagnosed with precision and could receive appropriate treatment at an earlier stage.”
The authors conclude with their future vision, “Our new IP/RT-QuIC assay may have many future applications as a biomarker for precise diagnosis and monitoring of treatment of neurodegenerative diseases in clinical trials. This simple diagnostic method will enable establishment of personalized therapy options for synucleinopathies.”
Funding
This study was supported by AMED under Grant Number JP19dm0207070 (Brain Mapping by Integrated Neurotechnologies for Disease Studies) to N.H., JP19dm0107156 (Strategic Research Program for Brain Sciences) to T.H. and A.O., JP21wm0425015 (Brain Mechanisms and Integrated Technologies for Mental Health and Disease Studies) to T.H. and A.O., JP19ak0101112 (Research on Development of New Drugs) to T.H. and N.H., JP21dk0207055 (Research and Development Grants for Dementia) to T.H., and JP16dm0107140 (Strategic Research Program for Brain Sciences) to N.N.; Grants-in-Aid for Scientific Research (21H04820 to NH, 21K07424 to T.H., 19K16928 to A.O., and 20K22693 to SU) from the Japan Society for the Promotion of Science; Visionary Council on the Moonshot Research and Development Program (JPMJMS2024-5 to N.H.) grants-in-aid from the Research Committee of CNS Degenerative Disease, Research on Policy Planning and Evaluation for Rare and Intractable Diseases, Health, Labor, and Welfare Sciences Research Grants; the Ministry of Health, Labor, and Welfare, Japan to N.H.; The Setsuro Fujii Memorial Osaka Foundation for Promotion of Fundamental Medical Research to T.H.; and Grants-in-Aid from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) of Japan to GM (17K07098 and 20H05333) for Scientific Research on Innovative Areas “Multimode autophagy.” The current work was also supported by the National Centre of Excellence in Research on Parkinson’s Disease (NCER-PD) funded by the Luxembourg National Research Fund (FNR; NCER13/BM/11264123) and the PEARL program (FNR; PEARL /P13/6682797 to RK and FNR and PEARL P16/BM/11192868 to MM).
Contact
For inquiries, please contact:
In relation to this study
Department of Neurology, Juntendo University School of Medicine Nobutaka Hattori, Professor, E-mail: nhattori “AT” juntendo.ac.jp
Taku Hatano, Senior Associate Professor, E-mail: thatano“AT”juntendo.ac.jp
TEL: +81-3-3813-3111
About the press release
Juntendo University School of Medicine
Yoshitaka Nakashima, E-mail: y.nakashima.gd “AT” juntendo.ac.jp
TEL: +81-3-5802-1590
Matters related to AMEDs projects
Division of Basic Medical Research, Department of Basic Medical Research
E-mail: brain-pm”AT”amed.go.jp
Telephone: +81-3-6870-2286
Replace “AT” with “@”
Reference
| Authors | Ayami Okuzumi1, Taku Hatano1, Gen Matsumoto2, Shuko Nojiri3, Shin-ich Ueno1, Yoko Imamichi1, Haruka Kimura1, Soichiro Kakuta4, Akihide Kondo5, Takeshi Fukuhara6, Yuanzhe Li1, Manabu Funayama1, Shinji Saiki1, Daisuke Taniguchi1, Taiji Tsunemi1, Deborah McIntyre7, Jean-Jacques Gérardy8, Michel Mittelbronn8, Rejko Krüger7, Yasuo Uchiyama9, Nobuyuki Nukina10, and Nobutaka Hattori1, 6 |
| Title of original paper | Propagative α-synuclein seeds in the serum of patients with synucleinopathies |
| Journal | Nature Medicine |
| DOI | 10.1038/s41591-023-02358-9 |
| Affiliations | 1Department of Neurology, Juntendo University School of Medicine, 2-1-1 Hongo, Bunkyo-ku, Tokyo 113-8421, Japan 2Department of Histology and Cell Biology, Nagasaki University School of Medicine, 1-12-4 Sakamoto, Nagasaki, 852-8523, Japan 3Medical Technology Innovation Center, Juntendo University Faculty of Medicine, 2-1-1 Hongo, Bunkyo-ku, Tokyo 113-8421, Japan 4Laboratory of Morphology and Image Analysis, Biomedical Research Core Facilities, Juntendo University Faculty of Medicine, 2-1-1 Hongo, Bunkyo-ku, Tokyo 113-8421, Japan 5The Department of Neurosurgery, Juntendo University Faculty of Medicine, 2-1-1 Hongo, Bunkyo-ku, Tokyo 113-8421, Japan 6Neurodegenerative Disorders Collaboration Laboratory, RIKEN Center for Brain Science, Saitama 351-0198, Japan 7Transversal Translational Medicine, Luxemburg Institute of Health (LIH); Centre Hospitalier de Luxemburg (CHL), Luxemburg; Translational Neuroscience, Luxemburg Centre for Systems Biomedicine (LCSB), University of Luxemburg 8Luxemburg National Center of Pathology (NCP), Laboratoire National de Sante (LNS), Dudelange, Luxemburg; Department of Cancer Research (DOCR), Luxemburg Institute of Health (LIH); Luxemburg Center of Neuropathology (LCNP), Luxemburg Centre for Systems Biomedicine (LCSB) Faculty of Science, Technology and Medicine (FSTM), and Department of Life Science and Medicine (DLSM), University of (LCSB) 9Department of Cell Biology and Neuroscience, Juntendo University Faculty of Medicine, 2-1-1 Hongo, Bunkyo-ku, Tokyo 113-8421, Japan 10Laboratory of Structural Neuropathology, Graduate School of Brain Science, Doshisha University, Kyoto 602-8580, Japan |